Enhancing T Cell Immunotherapy with CRISPR
From Gene Editing to Functional Reprogramming
CRISPR-Powered T Cell Immunotherapy
From Gene Editing to Functional Reprogramming of Immune Responses
In recent years, immunotherapy has emerged as a transformative approach in cancer treatment, with T cell–based strategies gaining particular attention. As key effectors of the adaptive immune system, T cells can recognize tumor-associated antigens and mediate cytotoxic responses, enabling precise elimination of abnormal cells. This intrinsic capability forms the foundation of T cell–based therapies.
However, in the complex tumor environment, T cell function is often significantly constrained:
● Tumor cells evade immune recognition by downregulating antigen expression or altering antigen presentation mechanisms
● Immunosuppressive signals within the tumor microenvironment, such as immune checkpoint activation (e.g., PD-1/PD-L1), inhibit T cell activation and cytotoxicity
● Chronic antigen stimulation can drive T cell exhaustion, characterized by reduced proliferation, impaired effector function, and diminished persistence
These factors collectively limit the therapeutic efficacy of native T cells.
To overcome these challenges, T cell engineering has become a key strategy to reshape immune function. With the advancement of CRISPR gene editing, T cell engineering has entered a more precise and programmable era.
Compared with conventional approaches, CRISPR enables targeted manipulation at endogenous genomic loci, offering enhanced controllability and flexibility for systematic T cell reprogramming. For example, multiplex editing strategies can simultaneously enhance antigen recognition, persistence, and resistance to immunosuppression—shifting from simple functional enhancement to comprehensive functional reprogramming.
Based on diverse gene editing strategies, EDITGENE provides gene knockout and knock-in services, along with robust and reliable cell models to support immune function research and cell engineering applications.
CRISPR-Based T Cell Editing
From Gene Disruption to Precision Reprogramming
Gene editing is the core technology enabling functional modulation in T cell engineering. CRISPR-based approaches include gene knockout, knock-in, and multiplex editing strategies tailored to different research and application needs.
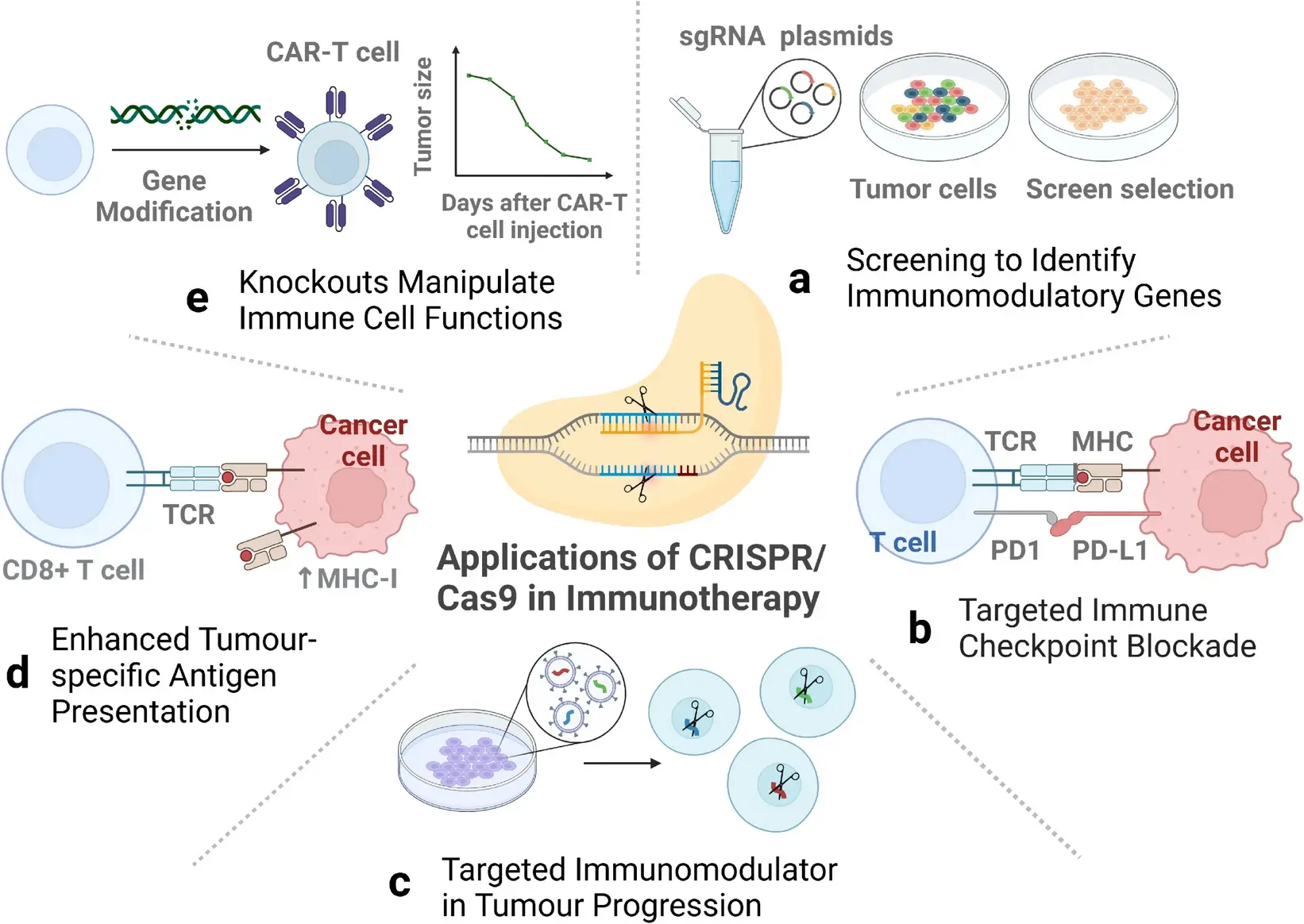
Feng et al., Exp Hematol Oncol, 2024
Gene Knockout (KO):
Targeted disruption of specific genes allows removal of negative regulators, thereby enhancing T cell activity.
Gene Knock-In (KI):
Site-specific insertion of functional elements (e.g., CAR constructs or regulatory sequences) expands T cell capabilities, enabling new antigen recognition or regulatory features. Compared with random integration, CRISPR-mediated targeted knock-in significantly improves expression stability and consistency.
Multiplex Editing:
Simultaneous modification of multiple genes enables combinatorial optimization—enhancing cytotoxicity, persistence, and resistance to immunosuppression within a single cell.
With over a decade of gene editing experience, EDITGENE has established four knock-in platforms—HES-KI, CRISPaint, Bingo™, and CRISPR/HDR—to meet diverse KI needs with high efficiency and precision.
Applications
● Identify key regulators by analyzing functional impacts of immune-related gene variants
● Construct precise knock-in models to study mutation or functional element effects
● Compare different gene modification strategies on T cell functions (cytotoxicity, persistence)
● Validate critical nodes in immune regulatory pathways
Services & Support
● Targeted knock-in (KI) model construction across multiple immune-related genes
● Custom solutions for precise engineering at endogenous loci
KO Cell Model in Immunology Research
From Baseline Activity to Enhanced Cytotoxicity and Persistence
Understanding how key genes regulate T cell function is essential for precise immune intervention.
CRISPR-mediated gene knockout provides a direct and efficient approach to dissect immune regulatory mechanisms by removing gene function at the genomic level. This enables systematic evaluation of gene roles in T cell activation, differentiation, and effector function.
Key Applications
● Knockout of immunosuppressive genes to enhance T cell activation and cytotoxicity
● Disruption of signaling pathway components to study proliferation, differentiation, and persistence
● Combinatorial knockout strategies to explore synergistic or antagonistic gene interactions
KO models also support comprehensive functional validation through multi-dimensional assays, including cytotoxicity analysis, cytokine profiling, and transcriptomics—linking gene regulation to functional phenotypes.
With a robust CRISPR platform, EDITGENE delivers efficient and stable knockout solutions for systematic T cell research.
What We Provide
● Single-gene and multi-gene knockout T cell models
● Customized KO design, screening, and validation workflows
● Reliable support for immune research and cell engineering optimization
Our Advantage:
A wide range of KO strategies—including frameshift mutations, fragment deletions, and multiplex knockouts—enables precise functional evaluation while minimizing background interference.
Overcoming Immunosuppressive Barriers
From Checkpoint Inhibition to Tumor Microenvironment Resistance
Although engineered T cells show strong activity in vitro, their function is often suppressed in vivo due to the tumor microenvironment (TME).
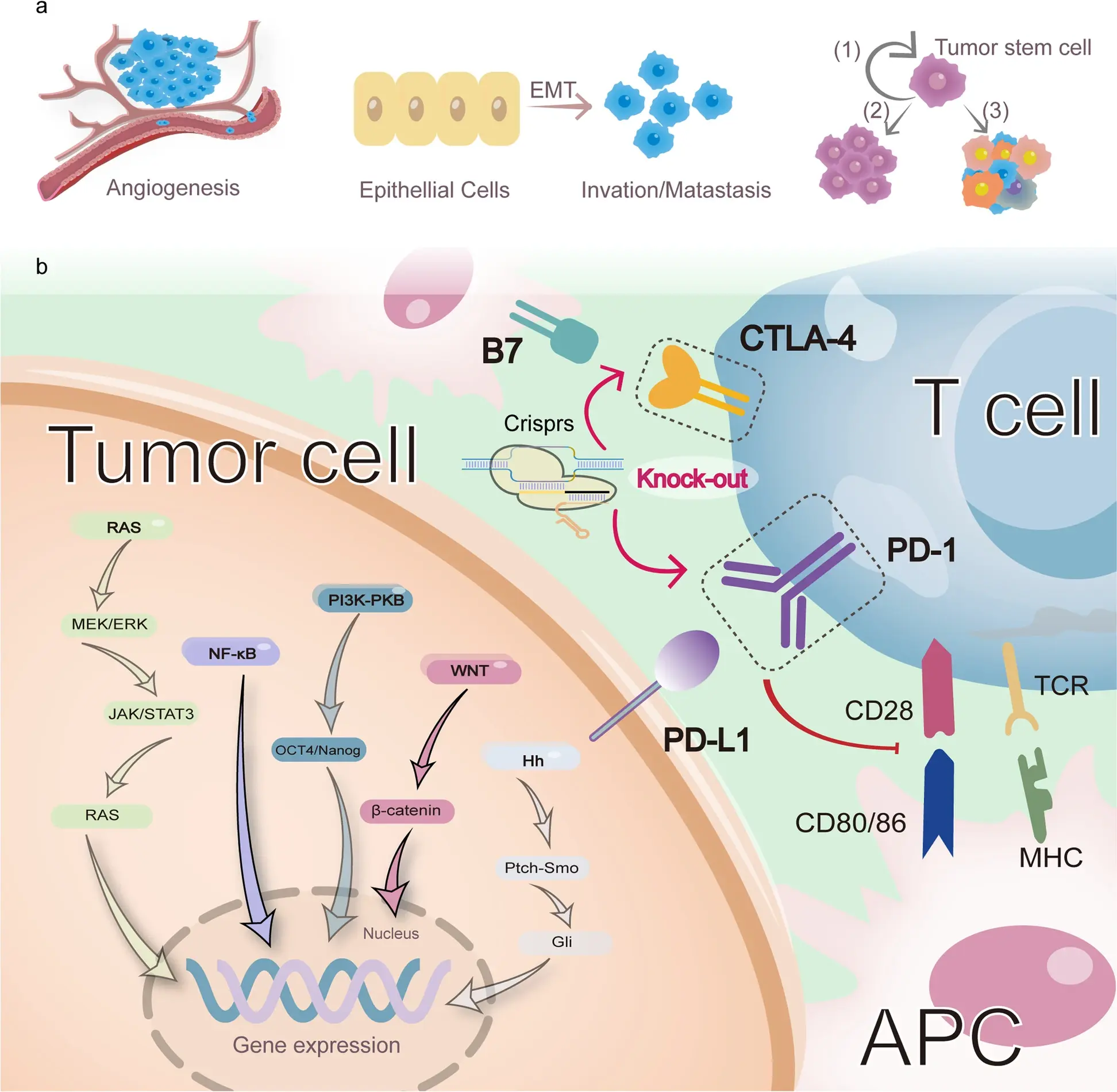
Liu et al., Mol Cancer, 2023
This environment consists of tumor cells, immunosuppressive cell populations, and cytokines that collectively inhibit T cell activation and function.
● Immune checkpoints (e.g., PD-1, CTLA-4) suppress proliferation and cytotoxicity
● Immunosuppressive cytokines (e.g., TGF-β, IL-10) and metabolic stress (hypoxia, nutrient competition) exacerbate T cell exhaustion
CRISPR provides both targeted and systematic solutions:
● Targeted Editing: Knockout of checkpoint genes restores T cell function
● CRISPR Screening: Identifies key regulators of metabolism and microenvironment adaptation
Genome-wide and targeted CRISPR screening enables systematic discovery of genes regulating T cell activity, proliferation, and persistence—supporting a full workflow from target discovery to functional validation and engineering optimization.
Applications:
● Identify regulators of T cell activity and immune modulation
● Discover pathways controlling proliferation, cytotoxicity, and persistence
● Reveal key suppressive factors in the tumor microenvironment
● Accelerate immunotherapy target discovery
CRISPR-Optimized CAR-T Strategies
From Conventional CAR Design to Precision-Engineered Cell Therapies
CAR-T therapy has shown remarkable success, particularly in hematological malignancies. However, traditional approaches rely on random integration, leading to variability in expression and safety concerns.
CRISPR enables:
● Site-specific CAR integration for improved stability and safety
● Multiplex editing to enhance antitumor activity, persistence, and resistance to suppression
● Transition from random engineering to precise, programmable cell therapy design
As CRISPR integrates with high-throughput screening and multiplex editing, next-generation CAR-T therapies are expected to achieve higher efficacy, safety, and adaptability.
End-to-End Workflow for T Cell Engineering
Immunosuppressive Mechanisms → Enhanced T Cell Function:
From immune recognition to identifying key targets that regulate T cell persistence and cytotoxicity, and further to achieving functional reprogramming through precise gene editing (knockout, knock-in, and multiplex editing), the EDITGENE platform provides end-to-end support for your research.
| Dimension |
Key Question |
Strategy |
Product/Service |
| Gene Knockout |
Which genes regulate or limit T cell function? |
CRISPR-mediated KO |
KO cell lines & custom KO services |
| Gene Knock-In |
How to introduce new functions? |
Targeted KI (CAR, mutations) |
Knock-in services |
| CRISPR Screening |
Which genes systematically control T cell activity? |
CRISPR library screening |
CRISPR libraries & screening services |
Platform Advantages
Built for Precision and Speed
Advanced Gene Editing Platforms
Integrating HES-KI and CRISPR-EDITx, our advanced platforms deliver efficient and reliable solutions for diverse gene editing needs.
Extensive KO Cell Line Library
Covering a wide range of genes, our extensive KO cell line library supports systematic loss-of-function studies across various disease models.
Fast Turnaround for Custom Models
With a turnaround time as short as 4 weeks for custom models, we significantly reduce project waiting time and accelerate your research.
Monoclonal Validation with Sequencing Confirmation
Monoclonal selection combined with sequencing confirmation ensures accuracy and reproducibility for every editing outcome.
Global Project Support and Delivery
Backed by a mature global project collaboration and logistics system, we provide full-process support from technical consultation to final delivery.
Experienced Team
Expert team with over 1000 gene editing projects and experience across 300+ cell types.























 Explore Cell Models
Explore Cell Models Request Project Design
Request Project Design Gene Knock-In Custom Service
Gene Knock-In Custom Service HES-KI Technology Platform
HES-KI Technology Platform Knockout Cell Models
Knockout Cell Models Customized Gene Knockout Services
Customized Gene Knockout Services