-
CRISPR Knockout KitCRISPR Point Mutation KitKI Enhancer Drug
-
Precision Mutation Cell PanelsWild Type Cell Line
-
-
 Knockout Cell Series - VEGF Signaling Pathway
Knockout Cell Series - VEGF Signaling Pathway
The Vascular Endothelial Growth Factor (VEGF) signaling pathway is a key regulator of angiogenesis, controlling endothelial cell growth, migration, and survival. Its dysregulation is closely linked to tumor angiogenesis, cardiovascular, and retinal diseases, making it a critical target for therapeutic research.
VEGF knockout cell lines enable precise pathway studies and disease modeling. Below, explore EDITGENE's validated VEGF pathway KO cell models and their key research applications.
The VEGF signaling pathway (Vascular Endothelial Growth Factor signaling pathway) is a central regulatory mechanism of angiogenesis, playing critical roles in both physiological and pathological processes.
Through activation of VEGF ligands and receptors, this pathway regulates the proliferation, migration, and survival of endothelial cells (ECs), enabling the controlled formation of new blood vessels during development, tissue repair, and regeneration.
However, dysregulation of VEGF signaling can lead to disease. Excessive VEGF activity promotes tumor growth, invasion, and metastasis by stimulating abnormal angiogenesis, whereas insufficient signaling can impair wound healing and contribute to ischemic disorders.
Because of its essential role in vascular biology and disease progression, the VEGF signaling pathway has become a major therapeutic target. A deeper understanding of VEGF pathway regulation is therefore critical for the development of anti-angiogenic therapies and novel disease treatments.
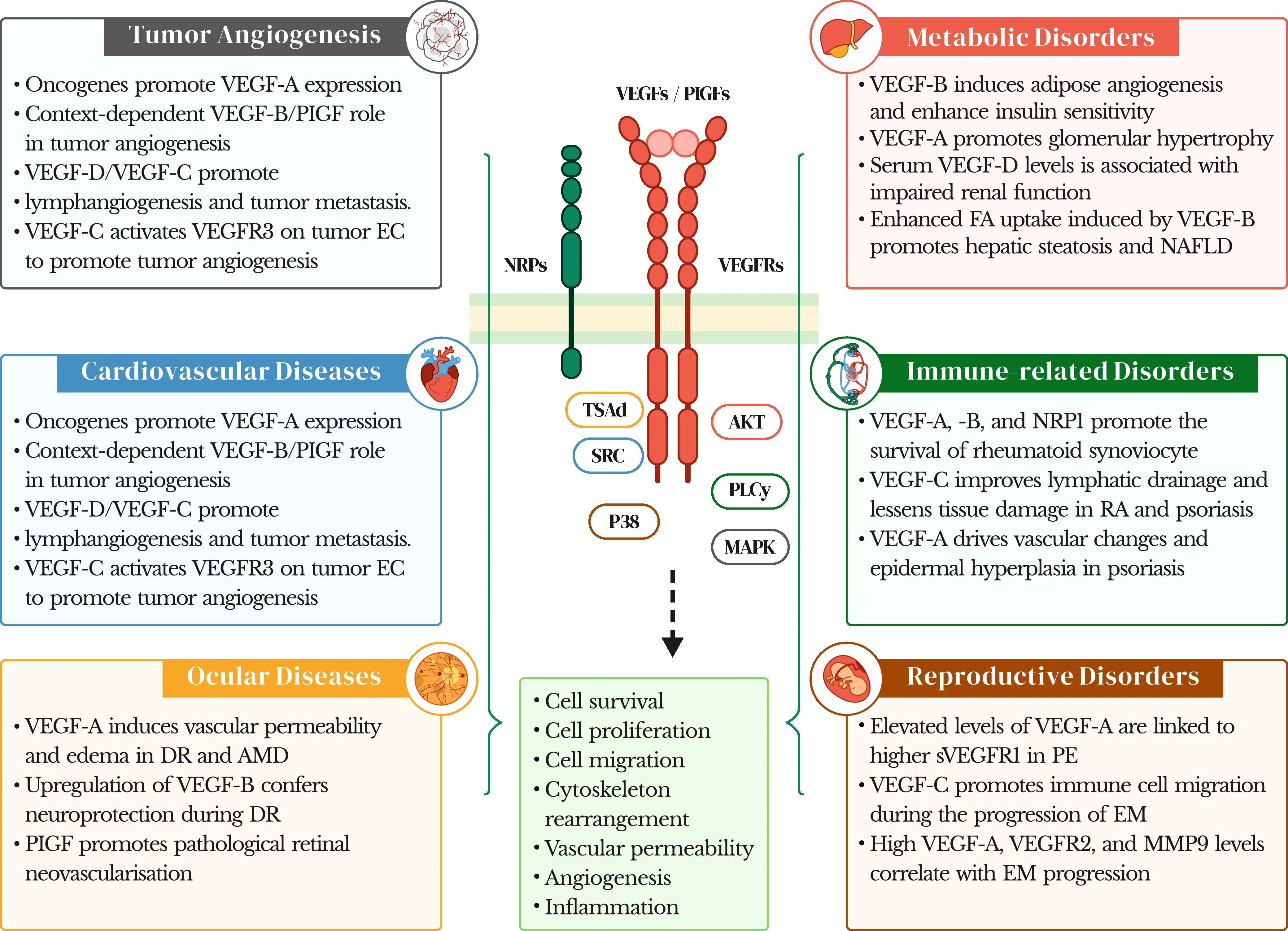
(Lee et al., Sig Transduct Target Ther, 2025)
VEGF ligands and their receptors play diverse roles in multiple pathological processes, including tumor angiogenesis, cardiovascular diseases, ocular disorders, and metabolic, immune, and reproductive diseases. Dysregulation of key molecular interactions and downstream signaling pathways can enhance cell survival, migration, and proliferation, contributing to diseases such as: Atherosclerosis (ATH), Myocardial infarction (MI), Diabetic retinopathy (DR), Age-related macular degeneration (AMD), Non-alcoholic fatty liver disease (NAFLD), Rheumatoid arthritis (RA), Preeclampsia (PE), Endometriosis (EM)
These disease contexts highlight the strong therapeutic potential of targeting the VEGF signaling pathway.
Gene knockout cell models provide powerful tools to investigate the regulatory functions of VEGF family members in different disease settings, supporting mechanistic studies, target validation, and drug discovery.
· Tumor & Angiogenesis Models
Investigate the roles of VEGF-A, VEGF-C, and related genes in tumor angiogenesis and lymphatic metastasis, and explore VEGFR3-mediated signaling mechanisms in tumor endothelial cells.
· Cardiovascular & Metabolic Disease Models
Analyze the inflammatory role of PlGF in atherosclerosis and study how VEGF-B regulates adipose angiogenesis and insulin sensitivity.
· Ophthalmic Disease Models
Develop VEGF-A–driven retinal disease models for studying diabetic retinopathy and macular degeneration, and explore the neuroprotective role of VEGF-B.
· Immune & Reproductive Disease Models
Examine the functions of VEGF-C in rheumatoid arthritis and endometriosis, and investigate the pathological mechanisms of VEGF-A in preeclampsia.
Explore the VEGF Signaling Pathway Knockout Cell Line Collection from EDITGENE, featuring validated models targeting key regulators of angiogenesis and vascular biology.
EDITGENE provides high-quality VEGF knockout cell lines for studying angiogenesis mechanisms, tumor vascularization, and VEGF-related disease pathways. Both in-stock and custom gene knockout cell lines are available to support diverse angiogenesis and vascular biology research needs.
-
Cat.No: EDC09412
species: Human
cell_name: HAP1
gene_name: TESK1
gene_id: 7016
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ251
species: Human
cell_name: HEK293
gene_name: VEGFC
gene_id: 7424
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ588
species: Human
cell_name: HEK293
gene_name: RELA
gene_id: 5970
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ724
species: Human
cell_name: HEK293
gene_name: PGF
gene_id: 5228
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ763
species: Human
cell_name: HEK293
gene_name: VEGFD
gene_id: 2277
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ1204
species: Human
cell_name: HEK293
gene_name: GAB1
gene_id: 2549
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ1314
species: Human
cell_name: HEK293
gene_name: DRD2
gene_id: 1813
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ2237
species: Human
cell_name: HEK293
gene_name: DRD4
gene_id: 1815
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ2726
species: Human
cell_name: HEK293
gene_name: DRD3
gene_id: 1814
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ3321
species: Human
cell_name: HEK293
gene_name: FOXC1
gene_id: 2296
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ3388
species: Human
cell_name: HEK293
gene_name: CSDE1
gene_id: 7812
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ5289
species: Human
cell_name: HEK293
gene_name: MYO1C
gene_id: 4641
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ5916
species: Human
cell_name: HEK293
gene_name: TESK1
gene_id: 7016
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ6377
species: Human
cell_name: HEK293
gene_name: NRP2
gene_id: 8828
specs: 1×10⁶cells
-
Cat.No: EDJ-KQ11479
species: Human
cell_name: HEK293
gene_name: GTPBP2
gene_id: 54676
specs: 1×10⁶cells
- 1
- 2
- Next Page »
Subscribe
You can unsubscribe from these communications at any time. For more information on how to unsubscribe, our privacy practices, and how we are committed to protecting and respecting your privacy, please review our Privacy Policy.
By clicking submit below, you consent to allow EDITGENE to store and process the personal information submitted above to provide you the content requested.