Common Transfection Methods for Gene Knockout and Their Comparison

Gene knockout refers to the process of inactivating or disrupting a specific gene within an organism’s genome. This technique has broad applications in life sciences, medicine, and drug development, including generating disease models, performing drug screening and target validation, and even achieving gene therapy by knocking out disease-causing genes.
Transfection is the forced introduction of small molecules such as DNA, RNA, or antibodies into eukaryotic cells. The commonly used methods for gene knockout include lentiviral transduction, chemical transfection (Lipofection), and electroporation (RNP delivery). Each method has its advantages and limitations, and their performance can vary depending on the experimental context.
Lentiviral transfection uses lentiviruses as vectors, which integrate their genome into transcriptionally active sites of the host genome through long terminal repeats (LTRs) at both ends. By replacing the sequence between the LTRs with a gene of interest, the modified virus can deliver this sequence into cells. Once integrated, the viral sequence—including the inserted gene—is stably maintained and expressed in daughter cells after host cell division.
One advantage of lentiviral transfection is that the Cas9 protein in a gene knockout system can be expressed separately in advance. This allows more flexibility for introducing other components, such as gRNAs, target genes, or donor templates. Performing knockout in a cell line with stably expressed Cas9 often yields higher editing efficiency than transient Cas9 transfection.
However, lentiviral transfection has some limitations. The introduced foreign genes integrate randomly into the genome, which may inadvertently affect the expression of other genes. The procedure is relatively long, the vector can only carry limited gene sizes, and the method requires more stringent experimental conditions.
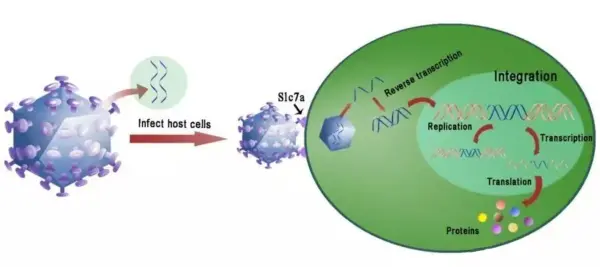
Lipid-mediated transfection, commonly using Lipofectamine, employs liposomes as carriers to encapsulate nucleic acids such as DNA or RNA within a hydrophilic core. The positively charged liposome surface interacts electrostatically with the negatively charged cell membrane, facilitating fusion with the membrane. The nucleic acids are then delivered into the cell via endocytosis.
Lipofection generally achieves high transfection efficiency, allowing the delivery of large amounts of nucleic acids into cells. The procedure is relatively straightforward and does not require specialized equipment or advanced technical skills. However, this method can be highly cytotoxic, making it less suitable for sensitive cells. Not all cell types are compatible with liposome-mediated transfection, and careful optimization is often necessary.
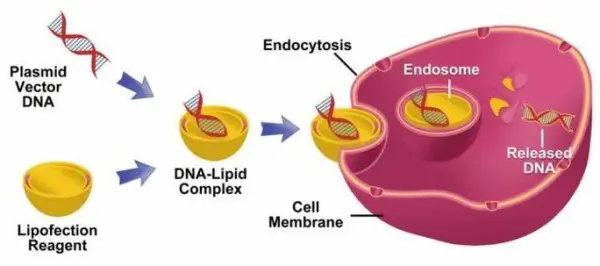
Electroporation is a physical transfection method that applies a brief, high-voltage electric pulse to target cells. This pulse temporarily forms nanoscale pores in the cell membrane, allowing genetic material, proteins, or other molecules to enter the cytoplasm. Once the pulse ends, the pores close, and the cells return to their normal state, completing the transfection process.
Electroporation can achieve highly efficient delivery, making it suitable for large molecules, negatively charged substances, or molecules that are otherwise difficult to introduce into cells. The procedure is relatively quick and reproducible, does not require a carrier, and is broadly applicable across cell types.
However, the high-voltage pulse can cause significant cellular damage. Only a fraction of cells successfully repair their membranes, resulting in substantial cell death. Compared with chemical transfection, electroporation typically requires more starting cells, and the parameters—such as voltage, resistance, and capacitance—must be carefully optimized to balance transfection efficiency with cell viability.
All three commonly used transfection methods have notable limitations: lentiviral transduction is time-consuming; lipofectamine can be highly cytotoxic and is limited to certain cell types; electroporation requires extensive parameter optimization and causes significant cell damage.
So, is there a method that can deliver transfection quickly, without cytotoxicity or cell damage, with broad applicability, and no need for extensive optimization? EDITGENE’s newly launched CRISPR EDITx™ KO Rapid Knockout Kit achieves exactly that!

The Flash Pro Rapid Knockout Kit leverages EDITGENE’s innovative CRISPR RNP delivery system along with Cas enzymes validated across thousands of experiments. DNA cleavage can be detected within 6 hours, and complete gene knockout is achieved in 48 hours, delivering true “fast, precise, and stable” performance to help you achieve accurate and efficient knockouts.Click here to view product details.
Specializes in CRISPR technology, EDITGENE is dedicated to providing comprehensive gene editing services and in vitro diagnostic products, including CRISPR library screening , cell-based gene editing , monoclonal cells screening and CRISPR-based detection .
EDITGENE is committed to delivering the most efficient technical solutions to support CRISPR-related research, gene function analysis, diagnostics, and therapeutic development.
Contact us
+ 833-226-3234 (USA Toll-free)
+1-224-345-1927 (USA)
info@editxor.com