SLC Transporter Knockout Cells: From Metabolic Regulation to Drug Target Discovery

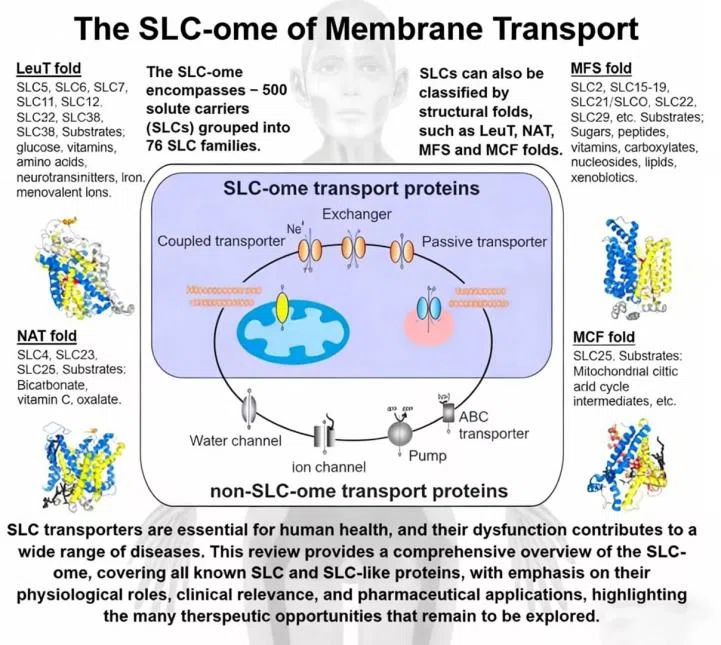
The Solute Carrier (SLC) superfamily is the largest group of transmembrane transporter proteins in the human body, with its encoding genes accounting for approximately 5.2% of the human protein-coding genome. From glucose and amino acids to drug molecules, SLCs regulate the transmembrane transport of virtually all essential substances for life.
In recent years, driven by advances in functional genomics and high-throughput screening technologies, SLC transporters have transitioned from being "underappreciated supporting roles" to core targets in the fields of metabolic diseases, oncology, and drug development. Gene knockout (KO) cell models have thus emerged as the gold-standard tools for elucidating SLC functions and validating these drug targets.
The SLC superfamily comprises approximately 460 members distributed across 76 families, along with 52 SLC-like proteins, bringing the total to 516. They act not only as the gatekeepers for nutrients entering and exiting the cell, but their dysfunction is also directly implicated in the pathogenesis of various major diseases.
CRISPR/Cas9-mediated gene knockout achieves complete loss of target gene function at the genomic level, offering irreplaceable advantages:
l Complete Loss of Function: Knockout leads to total protein inactivation, yielding clear phenotypes and high reproducibility.
l Stable Inheritance: Monoclonal cell lines can be passaged long-term without the need for repeated transfections.
l Suitability for High-Throughput Screening: They can be utilized to construct SLC family knockout libraries for systematic genetic interaction profiling.
A 2025 study published in Molecular Systems Biology mapped the large-scale genetic interactions of the human SLC superfamily using the CRISPR system. By screening 35,421 pairs of SLC-SLC double knockouts, researchers discovered that cellular transport systems exhibit remarkable metabolic redundancy and robustness, with only 0.67% of SLC-SLC gene pairs displaying genetic interactions.
This finding suggests that targeting a single SLC may be insufficient to produce a distinct phenotype, necessitating combinatorial knockouts or functional synergy analyses.
Beyond knockout strategies, point mutation models are another crucial tool for deciphering SLC functions. Many disease-associated SLC mutations are missense mutations (single amino acid substitutions) rather than a complete loss of function. Through CRISPR-mediated precise point mutations, researchers can:
l Model specific SLC missense mutations found in human genetic diseases.
l Investigate the impact of critical amino acid residues on transport activity and substrate selectivity.
l Screen for specific inhibitors targeting mutant variants.
1. SLC38A1: A Novel Metabolic Therapeutic Target in MDS/AML
SLC38A1 is a non-classical glutamine transporter.
A study presented at the 2025 AACR Annual Meeting revealed that SLC38A1 is significantly overexpressed in the long-term hematopoietic stem cells of patients with high-risk myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML), which correlates with shortened overall survival.
Utilizing CRISPR-mediated SLC38A1 knockout, researchers demonstrated a 37%, 42%, and 43% reduction in surviving colonies in MOLM13, THP-1, and MDS-L cells, respectively. Similarly, knockout effectively suppressed cell viability and colony-forming capacity in primary samples from AML and high-risk MDS patients.
This study indicates that metabolic intervention strategies targeting SLC38A1 hold promise for providing new therapeutic options for MDS/AML patients.
2. SLC22A6 (OAT1): A Classic Model for Renal Drug Transport
Organic anion transporter 1 (OAT1/SLC22A6) is expressed in renal proximal tubule cells, mediating the uptake of numerous clinical drugs, including antivirals, antibiotics, and diuretics. Loss of OAT1 function can lead to decreased drug clearance, heightening the risk of toxicity.
By leveraging SLC22A6 knockout cell models, researchers can evaluate whether candidate compounds are OAT1 substrates, predict drug-drug interactions, and screen for new molecular entities with low nephrotoxicity.
Genome-wide CRISPR screening has confirmed that SLC35A1 knockout significantly inhibits viral replication.
Furthermore, knocking out SLC35A1 in HEK293T and HeLa cells enhances the transduction efficiency of AAV9, providing an optimized tool for testing gene therapy efficacy.
Off-the-Shelf SLC KO/Point Mutation Cell Libraries
Product Advantages:
Partial List of Off-the-Shelf SLC Knockout Cells
The SLC transporter family is stepping out of the "supporting" shadows in basic research to become the "leading star" in drug target discovery. Whether exploring tumor metabolic reprogramming, deciphering drug transport mechanisms, or developing novel therapeutic strategies, SLC knockout cell models remain an indispensable core tool.
References
2. Alam, S., et al. (2023). Membrane transporters in cell physiology, cancer metabolism and drug response. Disease Models & Mechanisms, 16(11), dmm050404.
3. Gou, X., et al. (2022). Construction and evaluation of a novel organic anion transporter 1/3 CRISPR/Cas9 double-knockout rat model. Pharmaceutics, 14(11), 2307.
4. Girardi, E., et al. (2020). A widespread role for SLC transmembrane transporters in resistance to cytotoxic drugs. Nature Chemical Biology, 16(4), 469–478.